background
For this project we were assigned groups to make a chemistry based board game. This was lightly based off of the Rube Goldberg project from Freshman year. In our game we had to incorporate multiple chemical and mechanical reactions throughout the length of the game ending by lighting an LED. As the players continued through the game they would have to follow the instructions to complete the reactions to move on. We also needed to have a list of instruction, safety precautions and blueprints of the board. We had about three to four weeks to design and build our games at which point we would present to parents and other student at the ¨family game night¨.
Our Project
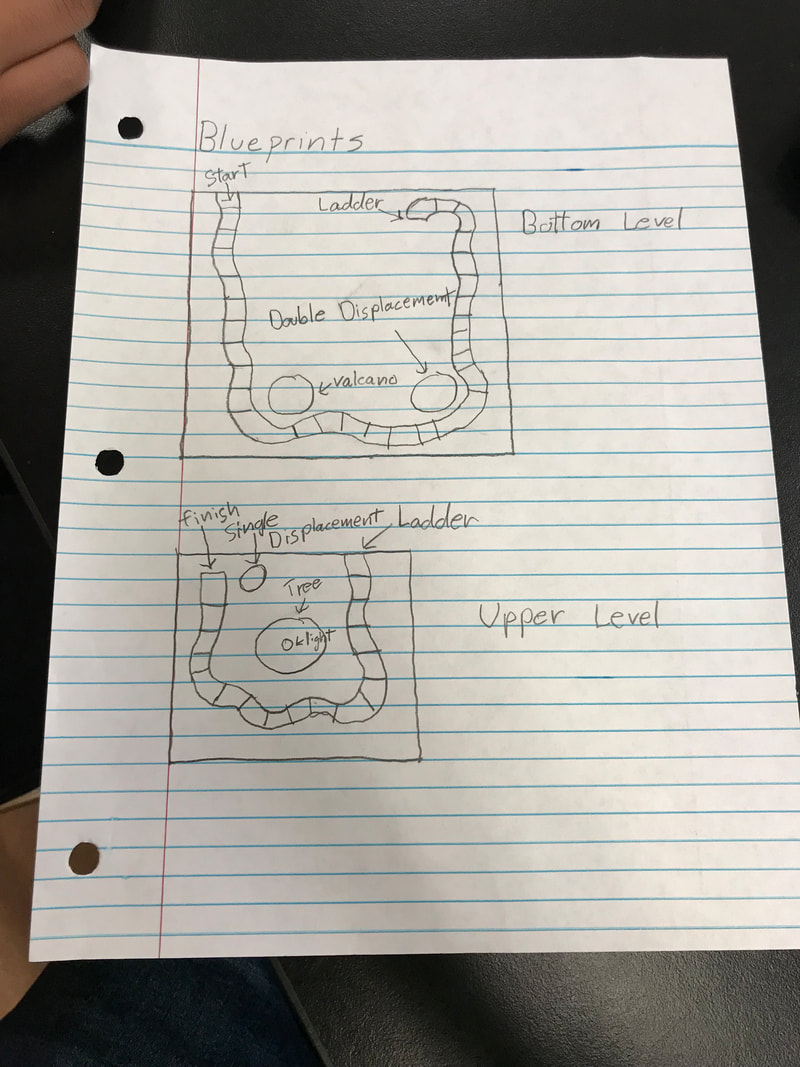
When my group and I were tasked with this project we started brainstorming ideas for our board game. After a while of talking about it we decided on a Christmas themed Candyland game. our next challenge was to find reactions for the game and a way to integrate them into the project smoothly. The basic way that you play the game of Candyland is by picking up cards with colors on them. When you get a color you move your piece to the next space with the color of the card on it. throughout the game there are special squares that when landed on require the player to complete ca chemical reaction before moving on. These places are where our single displacement, double displacement, gas production, and LED lighting reactions took place. Our first reaction was a gas production, the player must combine Sodium Hydrogen Carbonate and acetic acid otherwise known as baking soda and vinegar in a Christmas volcano to create gas. Next the double displacement reaction occurs when a player combines combines potassium iodide and lead nitrate turning the mixture yellow, creating the lemon candy for the Candyland. Lastly for the single displacement reaction the player must add Copper Nitrate to a dish soaking a strip of aluminum foil. The foil was attached to a breadboard that was connected to the LED at the top of the tree. The copper nitrate dissolves the aluminum foil completing the electrical circuit and lighting the LED.
Here are some pictures of our game
And here is our Instructions for the board game.
Blueprint
Important Concepts
Single Replacement: A type of chemical reaction where an element reacts with a compound and takes the place of another element in that compound
Double Replacement: A type of chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products
LED: A light-emitting diode is a two-lead semiconductor light source
Energy: The property that must be transferred to an object in order to perform work on, or to heat, the object
Physical Change: Changes affecting the form of a chemical substance, but not its chemical composition
Board Game: A tabletop game that involves counters or pieces moved or placed on a pre-marked surface or "board", according to a set of rules
Element: An element is a piece of matter that is made up of only one type of atom. An example would be iron, which is made up of only iron atoms
Compound: A compound is a molecule made up of multiple elements, so it has more than one type of atom. An example would be acetate, which is made of two carbon, three hydrogen, and two oxygen atoms
Cation: A cation is an element or compound that has more protons than electrons. Metals are almost always cations
Anion: An element or compound that has more electrons than it has protons. Anions have a negative charge. Non-metals are generally anions, with the notable exceptions of ammonium and hydronium
Synthesis Reaction: A synthesis reaction is when two or more elements or compounds all combine to form only one product. If a reaction has a single product, it must be a synthesis reaction.
Decomposition Reaction: A decomposition reaction is when one compound breaks apart into two or more elements or compounds. It is basically the opposite of a synthesis reaction, with only one reactant and multiple products.
Double Replacement: A type of chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products
LED: A light-emitting diode is a two-lead semiconductor light source
Energy: The property that must be transferred to an object in order to perform work on, or to heat, the object
Physical Change: Changes affecting the form of a chemical substance, but not its chemical composition
Board Game: A tabletop game that involves counters or pieces moved or placed on a pre-marked surface or "board", according to a set of rules
Element: An element is a piece of matter that is made up of only one type of atom. An example would be iron, which is made up of only iron atoms
Compound: A compound is a molecule made up of multiple elements, so it has more than one type of atom. An example would be acetate, which is made of two carbon, three hydrogen, and two oxygen atoms
Cation: A cation is an element or compound that has more protons than electrons. Metals are almost always cations
Anion: An element or compound that has more electrons than it has protons. Anions have a negative charge. Non-metals are generally anions, with the notable exceptions of ammonium and hydronium
Synthesis Reaction: A synthesis reaction is when two or more elements or compounds all combine to form only one product. If a reaction has a single product, it must be a synthesis reaction.
Decomposition Reaction: A decomposition reaction is when one compound breaks apart into two or more elements or compounds. It is basically the opposite of a synthesis reaction, with only one reactant and multiple products.
Reflections
This project was super fun and interesting, it was a new experience and an interesting way to learn about chemical reactions. My group mates and I worked very well together but near the beginning of the project after we had come up with a plan and started the build process I had to go on a trip to Argentina for a family trip. Sadly I was gone for the rest of the building of the project but I landed back in the US the same day as the ¨Family game night¨ so I showed up to help present to the parents. If I could go back I would have loved to help more with the project and not put so much work on my group. One thing I did well this project while I was here was that I had good work ethic. Something I need to improve on is my leadership. One of my highs was being there for the first part of the building, and a low was missing most of the project.